Current and Future Treatments of EoE
The standard approach to EoE treatment consists of the “3Ds:” diet, drugs, and dilation.1,2 Once EoE is diagnosed, shared decision making provides an excellent opportunity to discuss treatment modalities and patient preferences and values. Refer to the treatment algorithm in Figure 1.3
Diet
The elimination of food allergy triggers that promote an inflammatory response in the esophageal mucosa is the goal of dietary management strategies.4 There are three main dietary approaches to eliminating potential triggers4:
- Elemental diet. This is the most effective approach at bringing about symptom remission, with a response rate of over 90%. This approach uses amino acid-based formulas (AAF) for intake, along with some treats. Elemental diet is efficacious but difficult to maintain.
- Empiric dietary elimination. This diet follows the six-food Elimination Diet (SFED): dairy, wheat, eggs, soy, nuts, and seafood. While less restrictive than the elemental diet, it achieves only a to 70% response rate. Due to the restrictive nature of the SEFD, versions have been created that eliminate one (dairy), two (dairy and wheat), or four foods (dairy, wheat, egg, soy).
Patients treated with elimination diets should be referred to a registered dietitian if they experience excessive weight change, poor adherence, poor nutritional quality, or a negative impact on quality of life.1 Poor adherence to elimination diets, such as the SEFD, is influenced by effectiveness, social situations, and diet-related anxiety.6
Pharmacologic Therapy
There are two traditional choices of drug class: proton pump inhibitors (PPI) and oral formulations of topical corticosteroids.7 An initial trial of PPI therapy was the standard of care until updated diagnostic guidelines in 2018. Since then they have been considered as one option for initial treatment.8 This approach is inexpensive and tolerable, with a favorable safety profile.7
Swallowed topical corticosteroids have been routinely used in the treatment of EoE, with budesonide oral suspension receiving approval in 2024 for a 12-week course of treatment in those a 12-week course and older with EoE.9 A systematic review of double-blind clinical trials demonstrated that patients treated with topical corticosteroids for 2 to 12 weeks have approximately 3 times the rate of histological remission compared with placebo-treated patients (RR 0.39, 95% CI, 0.85 – 1.19). Topical steroids are well tolerated although Candida infections (12% to 15% of patients) can be a complication. There is no indication that adrenal insufficiency is a common side effect of topical steroid treatment (0.9%).10
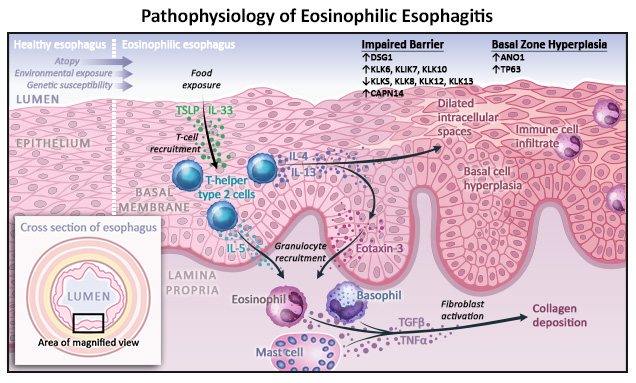
Newer, targeted agents show promise for treating patients with EoE due to the type 2 (Th2) inflammation that drives its pathophysiology. In June of 2022, the FDA approved the first biologic, dupilumab, for use in patients with EoE who are 12 years and older.11 Dupilumab has since received expanded approval in 2024, with approved indications for use in patients 1 year and older weighing at least 15 kg.12 It binds to the interleukin (IL)-4 receptor α and suppresses the action of Th2 and type 2 innate lymphoid cells (ILC2) in response to IL-4 and IL-13 (Figure 2).7 Please see the animations using the menu for a more in-depth discussion of EoE pathophysiology and targeted agents.
Numerous other agents are currently under investigation as therapy for patients with EoE.13 Table 1 lists the EoE drugs under investigation and their current status.

Dilation When EoE and inflammation have been ongoing for some time, esophageal strictures and narrowing may occur, leading to dysphagia and food impaction.7 Esophageal dilation can be successfully used to treat strictures but is normally used in patients after other treatments have failed. Studies do not support previous concerns regarding a high risk of complications, such perforation or ripping. 14 Patients most likely to benefit from dilation include: 14
- Those with strictures identified endoscopically with symptoms of dysphagia
- Those presenting with dysphagia symptoms in the absence of endoscopically apparent stricture
- Those who avoid symptoms of dysphagia because of adaptive eating behaviors and food avoidance, despite the presence of high-grade esophageal strictures (<10 mm)
References
- Shah A, Hirano I. Treatment of eosinophilic esophagitis: Drugs, diet, or dilation? Curr Gastroenterol Rep. 2007;9(3):181-8. doi:10.1007/s11894-007-0016-1
- Chen JW. Management of eosinophilic esophagitis: Dietary and nondietary approaches. Nutr Clin Pract. 2020;35(5):835-847. doi:10.1002/ncp.10571
- Gupta M, Grinman M. Diagnosis and management of eosinophilic esophagitis. CMAJ. 2024;196(4):E121-E128.
- Durban R, Dellon ES. Nutritional care of the patient with eosinophilic esophagitis. Practical Gastroenterol. 2018;17(4):40-51. https://practicalgastro.com/2019/07/30/nutritional-care-of-the-patient-with-eosinophilic-esophagitis
- Falk GW, Pesek R. Pharmacologic management of eosinophilic esophagitis. Immunol Allergy Clin N Am. 2024;44:245-264.
- Wang R, Hirano I, Doerfler B, et al. Assessing adherence and barriers to long‑term elimination diet therapy in adults with eosinophilic esophagitis. Dig Dis Sci. 2018;63(7):1756-1762. doi:10.1007/s10620-018-5045-0
- Muir A, Falk GW. Eosinophilic esophagitis: A review. JAMA. 2021;326(13):1310-1318. doi:10.1001/jama.2021.14920
- Budesonide oral suspension (EohiliaTM) Prescribing Information (PI) 2025. (https://content.takeda.com/?contenttype=PI&product=EOH&language=ENG&country=USA&documentnumber=1)
- Dellon ES, Liacouras CA, Molina-Infante J, et al. Updated international consensus diagnostic criteria for eosinophilic esophagitis: Proceedings of the AGREE conference. Gastroenterol. 2018;155(4):1022-1033.e10. doi:10.1053/j.gastro.2018.07.009
- Hirano I, Chan ES, Rank MA, et al. AGA Institute and the Joint Task Force on Allergy-Immunology Practice Parameters Clinical Guidelines for the Management of Eosinophilic Esophagitis. Gastroenterol. 2020;158(6):1776-1786.doi:10.1053/j.gastro.2020.02.038
- News Release 5/20/22 (https://www.prnewswire.com/news-releases/fda-approves-first-treatment-for-eosinophilic-esophagitis-a-chronic-immune-disorder-301552266.html)
- Dupilumab (Dupixent®) PI 2025 (https://www.regeneron.com/downloads/dupixent_fpi.pdf)
- Khokhar D, Marella S, Idelman G, et al. Eosinophilic esophagitis: Immune mechanisms and therapeutic targets [published online ahead of print, 2022 Jul 1]. Clin Exp Allergy. 2022;10.1111/cea.14196. doi:10.1111/cea.14196
- Katzka DA. Esophageal dilation as the primary treatment for eosinophilic esophagitis. Gastroenterol Hepatol. 2019;15:320-322.
All URLs accessed 1/6/26.