GI Specialist Clinical Toolkit
Gastroenterologists play a central role in recognizing disease patterns, establishing diagnosis through endoscopy and biopsy, guiding treatment selection, and monitoring long-term outcomes. Advances in understanding EoE pathophysiology and the expansion of therapeutic options have reinforced the importance of early diagnosis and sustained disease control to prevent progression to fibrostenotic disease.¹,² Chronic, inadequately treated inflammation contributes to tissue remodeling, subepithelial fibrosis, and eventual development of rings and strictures—features more commonly observed in adults due to prolonged disease duration.3,4
Clinical Presentation and Diagnostic Approach
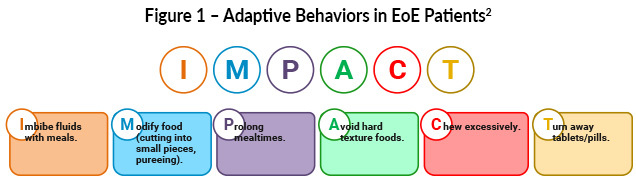
Symptoms of EoE vary by age and disease duration. Adults most commonly present with dysphagia, food impaction, heartburn, or chest discomfort, while pediatric patients may exhibit feeding difficulties, vomiting, or abdominal pain. Adaptive eating behaviors—such as excessive chewing, food avoidance, or prolonged meal times—can mask disease severity and should be actively assessed during evaluation using the IMPACT acronym (Figure 1).2,5
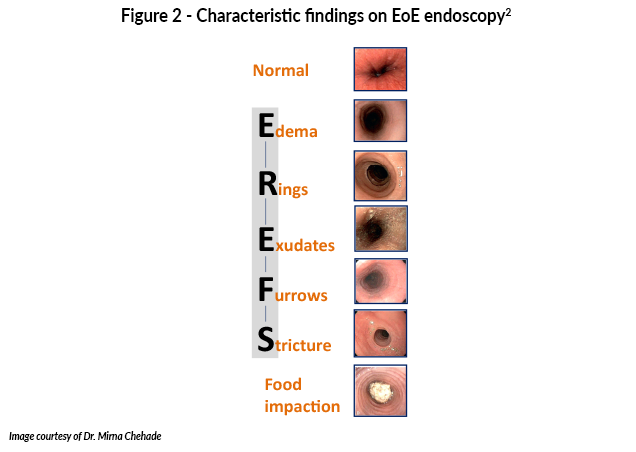
Histologic assessment with esophagogastroduodenoscopy (EGD) along with an eosinophil count of ≥15 eosinophils per high-power field on at least six biopsies from two or more esophageal levels is used to establish diagnosis.1 Figure 2 shows characteristic findings on endoscopy in EoE patients. Adult patients have significantly higher rates of esophageal strictures than pediatric patients with EoE, largely due to prolonged, untreated inflammation leading to progressive fibrosis.6 The most common differential diagnosis for EoE in adults is gastroesophageal reflux disease (GERD), as both conditions can present with similar symptoms and esophageal eosinophilia. Failure to respond to proton pump inhibitors (PPIs) is no longer used to distinguish GERD from EoE, as PPIs have an anti-inflammatory effect in EoE as well.1 Differentiating EoE from gastroesophageal reflux disease can be challenging due to overlapping clinical features.1
Clinical Presentation and Diagnostic Approach
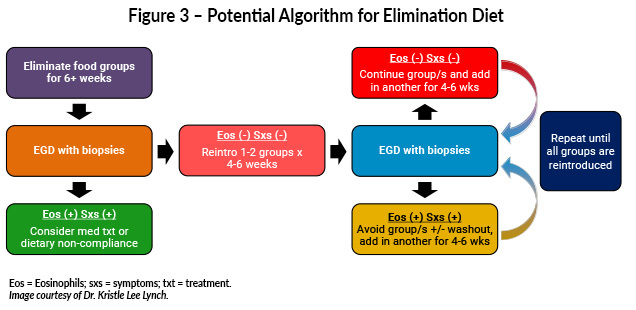
Empiric elimination diets are commonly used in adults and may be implemented through step-up or top-down approaches to balance efficacy with adherence (Figure 3).1 Elemental diets achieve high rates of histologic remission but are rarely used in adults due to poor palatability and significant treatment burden.1,7,8 Nutritional monitoring and collaboration with dietitians are essential when dietary therapy is employed.1,2
Pharmacologic therapy remains a cornerstone of EoE management. Proton pump inhibitors are frequently used as first-line treatment due to favorable safety, accessibility, and demonstrated anti-inflammatory effects.5 A recent meta-analysis of 73 studies including 7304 patients revealed that PPI therapy induces clinical and histological remission in almost half of the patients with EoE.9 Long-term data from a four-year phase 3 open-label study of the swallowed topical corticosteroid budesonide oral suspension demonstrated sustained histologic response in approximately half of patients aged 11 years and older, with maintained endoscopic improvement and similar outcomes in children and adults.10 Fluticasone propionate, another swallowed topical corticosteroid, has been studied in EoE using an investigational dispersible tablet formulation (APT-1011) designed to disintegrate and coat the esophageal mucosa for targeted topical delivery. In a phase 2b study of adults with EoE, fluticasone dispersible tablets demonstrated strong histologic efficacy at 12 weeks, with response rates of 80%–86% in twice-daily dosing groups compared with no response in the placebo group, and were generally well tolerated.11 While these results supported fast-track designation and further clinical development, fluticasone dispersible tablets are not currently FDA-approved or commercially available for EoE. Adrenal insufficiency is not considered a frequent adverse effect of swallowed corticosteroid therapy.2 Repeat endoscopy with biopsies is generally performed after initiation or modification of therapy, most commonly after approximately 8–12 weeks, to assess histologic response, regardless of treatment modality.1
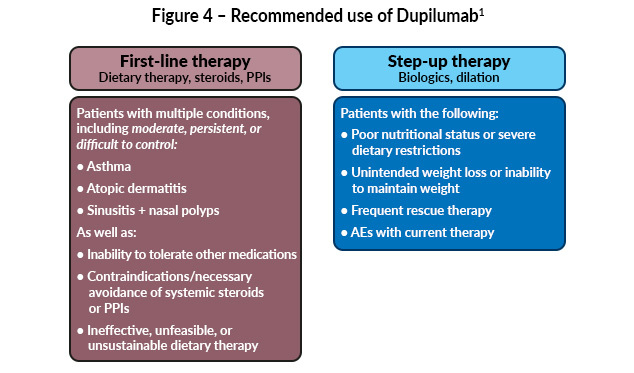
Targeted biologic therapy, dupilumab, was approved for adolescents aged ≥12 years in 2022 and expanded in 2024 to include children aged ≥1 year weighing at least 15 kg.12,13 Dupilumab may be considered as a second-line or subsequent therapy for patients with persistent disease despite dietary modification and/or swallowed topical corticosteroids. In select cases, such as patients with significant comorbid atopic diseases including asthma, atopic dermatitis, poor tolerance of dietary restrictions, or contraindications to steroids, it may be used earlier in the treatment course through shared decision-making.1
Esophageal dilation is reserved for patients with long-standing disease who develop fibrostenotic complications such as strictures or narrowing, which may result in dysphagia or food impaction.5 Esophageal dilation can be successfully used to treat strictures but is normally used in patients after other treatments have failed.14 Studies do not support previous concerns regarding a high risk of complications, such as perforation or ripping. Patients most likely to benefit from dilation include:14
- Those with strictures identified endoscopically with symptoms of dysphagia
- Those presenting with dysphagia symptoms in the absence of endoscopically apparent stricture
- Those who avoid symptoms of dysphagia because of adaptive eating behaviors and food avoidance, despite the presence of high-grade esophageal strictures (<10 mm)
Multidisciplinary Care of EoE Patients
Optimal management of EoE is accomplished through a multidisciplinary approach, involving not only gastroenterologists but also allergists/immunologists in patients with comorbid atopic diseases, registered dietitians or nutritionists, and primary care providers.⁵-8 Multidisciplinary care supports comprehensive assessment and monitoring, facilitates individualized therapeutic planning, and helps address the nutritional, immunologic, and psychosocial aspects of EoE that are important for adherence and better outcomes.15-18
Resources
Clinical guidelines and education for EoE management.
American College of Gastroenterology (ACG). https://gi.org
Practical tools for EoE diagnosis and treatment.
American Gastroenterological Association EoE Clinical Toolkits. https://gastro.org/clinical-guidance/guideline-toolkits/eosinophilic-esophagitis-toolkit/
Allergy-focused education on EoE.
American Academy of Allergy Asthma & Immunology (AAAI). https://www.aaaai.org/tools-for-the-public/conditions-library/allergies/eosinophilic-esophagitis
Patient-friendly information on EoE.
American Gastroenterological Association (AGA) Gastrointestinal Center. Eosinophilic esophagitis (EoE). https://patient.gastro.org/eosinophilic-esophagitis/
Advocacy, education, and support for eosinophilic disorders.
American Partnership for Eosinophilic Disorders (APFED). https://apfed.org/
Overview of the six-food elimination diet for EoE.
American Gastroenterological Association (AGA) Gastrointestinal (GI) Patient Center. Six-food Elimination Diet (SFED). https://patient.gastro.org/six-food-elimination-diet-sfed/
Global education and research for eosinophilic disorders.
EOSNETWORK. https://www.eosnetwork.org/
Directory of EoE-experienced providers.
EOSNETWORK. https://www.eosnetwork.org/find-a-doctor
Awareness and educational resources for EoE.
EoE Day Alliance. https://eoeday.org/resources/
Patient education on EoE and allergic disease overlap.
Asthma and Allergy Foundation of America. https://aafa.org/programs/education-programs/asthma-allergies-patient-caregiver-webinars/eosinophilic-esophagitis-eoe-webinars/
EoE education for patients and families.
SeeEoE. https://www.seeeoe.com/resources#resources-for-patients
Database of EoE clinical trials.
National Institutes of Health (NIH). US National Library of Medicine. ClinicalTrials.gov. https://www.clinicaltrials.gov
All URLs accessed on January 13, 2026
References
- Dellon ES, Muir AB, Katzka DA, et al. ACG clinical guideline: Diagnosis and management of eosinophilic esophagitis. Am J Gastroenterol. 2025;120:31-59. doi:10.14309/ajg.0000000000003194
- Hirano I, Furuta GT. Approaches and challenges to management of pediatric and adult patients with eosinophilic esophagitis. Gastroenterology. 2020;158:840-851. doi:10.1053/j.gastro.2019.09.052
- Dellon ES, Kim HP, Sperry SLW, et al. A phenotypic analysis shows that eosinophilic esophagitis is a progressive fibrostenotic disease. Gastrointest Endosc. 2014;79:577-585.e4. doi:10.1016/j.gie.2013.10.027
- Schoepfer AM, Safroneeva E, Bussmann C, et al. Delay in diagnosis of eosinophilic esophagitis increases risk for stricture formation. 2013;145:1230-1236.e62. doi:10.1053/j.gastro.2013.08.015
- Muir A, Falk GW. Eosinophilic esophagitis: A review. JAMA. 2021;326:1310-1318. doi:10.1001/jama.2021.14920
- Hirano I. Clinical relevance of esophageal subepithelial activity in eosinophilic esophagitis. J Gastroenterol. 2020;55:249-260. doi:10.1007/s00535-019-01624-3
- Shah A, Hirano I. Treatment of eosinophilic esophagitis: Drugs, diet, or dilation? Curr Gastroenterol Rep. 2007;9:181-188. doi:10.1007/s11894-007-0016-1
- Chen JW. Management of eosinophilic esophagitis: Dietary and nondietary approaches. Nutr Clin Pract. 2020;35:835-847. doi:10.1002/ncp.10571
- Lucendo AJ, Gutiérrez-Ramírez L, Tejera-Muñoz A, et al. Proton pump inhibitors for inducing and maintaining remission in eosinophilic esophagitis: An updated systematic review and meta-analysis. Clin Gastroenterol Hepatol. 2025;23:2115-2127.e21. doi:10.1016/j.cgh.2025.01.016
- Dellon ES, Katzka DA, Mukkada VA, et al. Long-term safety and efficacy of budesonide oral suspension for eosinophilic esophagitis: A 4-year, phase 3, open-label study. Clin Gastroenterol Hepatol. 2025;23:2155-2166.e5. doi:10.1016/j.cgh.2024.12.024
- Dellon ES, Lucendo AJ, Schlag C, et al. Orally disintegrating tablet (APT-1011) for eosinophilic esophagitis: randomized eontrolled trial. Clin Gastroenterol Hepatol. 2022;20:2485-2494.e15. doi:10.1016/j.cgh.2022.02.013
- News Release 5/20/22 (https://www.prnewswire.com/news-releases/fda-approves-first-treatment-for-eosinophilic-esophagitis-a-chronic-immune-disorder-301552266.html).
- Dupilumab (Dupixent®). Prescribing information. RegeneronPharmaceuticals, Inc; 2025. (https://www.regeneron.com/downloads/dupixent_fpi.pdf).
- Katzka DA. Esophageal dilation as the primary treatment for eosinophilic esophagitis. Gastroenterol Hepatol (N Y). 2019;15:320-322. PMCID: PMC6676355
- Dellon ES, Gonsalves N, Hirano I, et al; American College of Gastroenterology. ACG clinical guideline: evidenced based approach to the diagnosis and management of esophageal eosinophilia and eosinophilic esophagitis (EoE). Am J Gastroenterol. 2013;108:679-692. doi:10.1038/ajg.2013.71
- Lucendo AJ, Molina-Infante J, Arias Á, et al. Guidelines on eosinophilic esophagitis: evidence-based statements and recommendations for diagnosis and management in children and adults. United European Gastroenterol J. 2017;5:335-358. doi:10.1177/2050640616689525
- Chawla K, Alabbas B, Sheth D, Papademetriou M. As easy as EoE: A novel and effective multidisciplinary approach to care of patients with eosinophilic esophagitis in the age of biologics. Dig Dis Sci. 2020;65(8):2196-2202. doi:10.1007/s10620-020-06366-4
- Sauer BG, West A, McGowan EC. Nutritional care multidisciplinary eosinophilic esophagitis care: A model for comprehensive patient-centered care through shared decision making between gastroenterology, allergy, and nutrition of the patient with eosinophilic esophagitis. Clin Gastroenterol Hepatol. 2021;19:2226-2229. doi:10.1016/j.cgh.2021.07.025
All URLs accessed on January 13, 2026